Forming metal into (Secrets of stronger metals revealed) the shapes required for various purposes can be performed in many ways, including forming, processing, rolling, and forging. These processes affect the size and shapes of small crystal grains, which contain most metals, be they steel, aluminum or other widely used metals and alloys.
Now, MIT researchers have been able to study exactly what happens when these nut crystals form during a severe deformation process, on the smallest scale, up to a few nanometers in diameter. New discoveries (Secrets of stronger metals revealed) could lead to better processing methods to produce better and more consistent properties such as hardness and hardness.
The new findings (Secrets of stronger metals revealed), made possible by detailed analysis of images from various high-performance imaging systems, are published today in the journal Nature Materials in an article by former MIT postdoc Ahmed Tiamiya (now an assistant professor at the University of Calgary); MIT professors Christopher Schuh, Keith Nelson and James LeBeau; former student Edward Pang; and current student Xi Chen.
“In the process of making a metal, you give it a specific structure, and that structure will determine its properties in operation,” Schuh said. In general, the smaller the grain size, the stronger the resulting metal. Efforts to improve strength and durability by reducing grain size “have become a common theme throughout metallurgy, in all metals, over the last 80 years,” he said.
Metallurgists have long used various empirical methods that have been developed to reduce the grain size in a piece of solid metal, usually by causing different types of stress by deforming it in some way. But it is not easy to reduce these grains.
The most important method is called recrystallization, in which the metal is deformed and heated. It makes a lot of small mistakes in the whole part, which is “very messy and everywhere,” said Schuh, a professor of metallurgy at Danae and Vasilis Salapatas.
If the metal is damaged and heated, then all defects can spontaneously form new crystal nuclei. “From this galactic juice defect to new new nuclear crystals. And because they’re still new nuclear, they’re getting very small,” leading to a multi-grain structure, Schuh explains.
What is unique about the new job, he said, is determining how the process works at very high speeds and minimum scales. While conventional metal forming processes, such as forging or sheet metal rolling, can be very fast, according to Shoe, this new analysis focuses on processes that are “many orders of magnitude faster.”
“We use a laser to emit metal particles at supersonic speeds. To say that it happens in the blink of an eye is an incredible understatement, because you can make thousands of them in the blink of an eye,” Schuh said.
Such a high-speed process is not just a laboratory curiosity, he said. “There are processes in industry where things happen at such a pace.” These include a high-speed authorization form; high energy metal powder grinding; and a method called cold spraying for shaping and coating. In their experiments, we “tried to understand that the recrystallization process is one of the stricter rates, and because the speeds are so high, no one has been able to dig up there before and systematically look at the process,” he said.
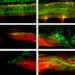
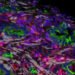
Tiamiyu, who conducted the experiments, using a laser system to fire 10 micron particles onto the surface, “was able to fire these particles at once and actually measure how fast they fly and how fast they move,” he says. Shoe. It shoots particles at higher speeds, then cuts them to see how the grain structure develops, up to the nanometer scale, using various sophisticated microscopic techniques at MIT.nano, with microscopy specialists.
The result was the discovery of what Schuh called a “new path” in which grains up to the nanometer scale are formed. The new path, which they call nano-twinning-assisted recrystallization, is a variation on a well-known phenomenon in metals called twinning, which is a special type of defect in which part of the crystal structure – inversion – is subject in its orientation. It’s a “mirror inversion of symmetry and you get these striped patterns where the metal reverses its orientation and repeats like a herringbone pattern,” he says. The team found that the higher the rate of these effects, the more this process occurs, leading to smaller grains as the nanoscale “twins” decompose into new crystalline grains.
In experiments they performed with copper, the process of bombarding the surface of these small particles at high speed could increase the strength of the metal by about ten times. “It’s not a small change in properties,” says Schuh, and this result is not surprising, as it is an increase in the well-known curing effect that comes from hammer blows in ordinary blacksmiths. . “He’s the kind of hypercaster we’re talking about.”
In the experiments, they were able to use a wide range of images and measurements of exactly the same particles and points of impact, says Schuh: “So we got a multimodal view. We take different lenses in the same exact area and material, and when you put them all together, you have a lot of detail about what happens that technology can’t provide.
Because new knowledge provides guidance on the desired level of deformation, how quickly the deformation will occur, and the temperatures to be used for maximum impact for particular metals or processing methods, they can be used directly to produce real metals. , sii Tiamiyu. The graphs they create from the experimental work should be widely available. “It’s not just hypothetical rules,” Tiamiyu said. For any given metal or alloy, “if you’re trying to figure out how to create a nanograin, if you have the parameters, just put them there” in the samples they create, and the results should indicate what kind of nut structure is possible. . expected at a given rate of influence and a given temperature.